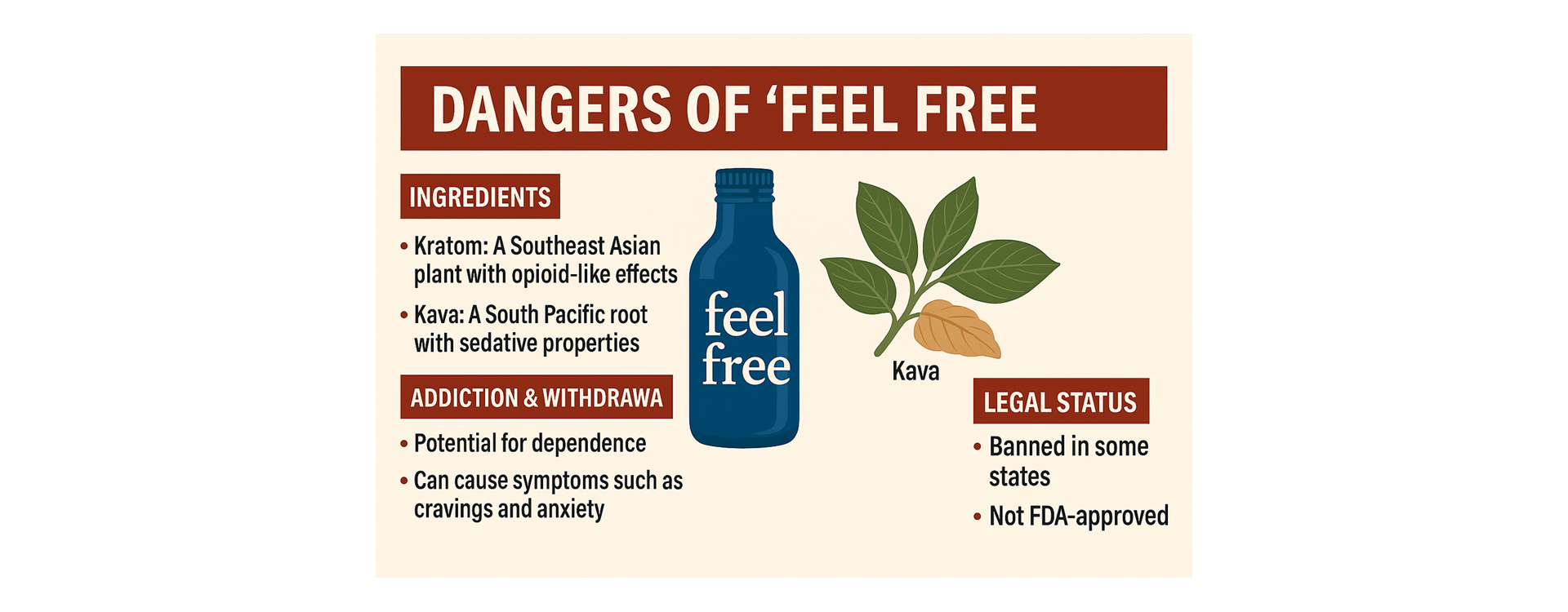
Marketed as a “plant-based wellness tonic” that promotes focus, energy, and relaxation, “Feel Free” by Botanic Tonics has rapidly gained popularity across the U.S. Its bright blue bottle and “natural” branding suggest a harmless herbal pick-me-up. But behind the glossy marketing lies a troubling truth: this drink’s key ingredients—kratom and kava—carry serious risks, including addiction, withdrawal, and potential organ toxicity [1][2].
“Feel Free” contains two psychoactive botanicals:
Together, they create sensations of relaxation, sociability, and focus. However, regular consumption can lead to dependence and withdrawal, particularly because kratom affects the same receptors as opioids [3].
Though marketed as a “natural” alcohol alternative, “Feel Free” has been compared by users and addiction specialists to prescription opioids for its potency and withdrawal symptoms. Many report developing cravings within days. The online community Reddit’s r/QuittingFeelFree grew from 200 to over 2,000 members in just one year, filled with stories of dependence, insomnia, anxiety, and even physical pain during withdrawal [2][4].
Some describe catastrophic effects. One Los Angeles man reported losing his job, apartment, and custody of his son while drinking up to 20 bottles per day, spending nearly $700 weekly. Others describe severe withdrawal symptoms such as muscle pain, nausea, sweats, and “freezing sensations” throughout the body. “It felt like ice in my veins,” said one recovering user, comparing the experience to heroin detox [2].
Kratom’s alkaloids bind to the same opioid receptors that control pain and pleasure, triggering the brain’s reward circuits and reinforcing dependency. Over time, this leads to tolerance and withdrawal symptoms similar to opioid addiction. Kava compounds may mask anxiety caused by withdrawal, making the cycle even harder to break [1].
While Botanic Tonics claims its drink uses whole-leaf kratom rather than extracts, users consistently report “Feel Free” as one of the most potent kratom-based products on the market, suggesting high concentrations of active compounds [2].
Beyond addiction, both ingredients carry well-documented risks:
Because “Feel Free” is sold as a dietary supplement, not a drug, it bypasses FDA safety regulations. The FDA has repeatedly warned about kratom’s potential for dependence, overdose, and contamination [1].
Critics argue that “Feel Free’s” branding intentionally appeals to people seeking sobriety or wellness by presenting it as a safe, “natural” alternative to alcohol. The drink has been heavily promoted on podcasts and social media as an aid for relaxation, energy, or creativity—without disclosing its addictive potential.
This is especially concerning for people in recovery, since kratom stimulates the same neurological pathways involved in opioid craving. Addiction treatment experts now warn that products like “Feel Free” can lead to cross-dependence and relapse [3][4].
As public scrutiny has increased, so has government and legal action:
Despite ongoing lawsuits, Botanic Tonics has not removed the product from shelves. It remains widely available at gas stations, smoke shops, and convenience stores across the country.
“Feel Free” exemplifies how easily “natural” marketing can disguise risk. Like nicotine or cocaine—both plant-based—kratom and kava are biologically active substances that alter brain chemistry. Phrases like “clean energy” and “Mother Nature’s wellness” obscure the reality of their addictive potential.
As one recovering user put it:
“It gives you something to lie to yourself about.”
While “Feel Free” promises focus and calm, its reality is far more dangerous. With growing evidence of addiction, physical harm, and regulatory gaps, the drink represents a cautionary tale in the wellness industry.
Consumers deserve transparency about what they’re putting into their bodies. True well-being isn’t found in another “miracle drink”—it starts with awareness, education, and informed choice.
Q: Is the Feel Free drink safe?
A: No. “Feel Free” contains kratom and kava, both of which can cause dependence, withdrawal, and health risks according to FDA warnings.
References
[1] U.S. Food and Drug Administration. FDA Advisories on Kratom Use and Safety Warnings (2018–2025).
[2] Punch Magazine. “The Little Blue Devil: Inside America’s Kratom Kava Craze” (July 16, 2024).
[3] Class-Action Lawsuit Against Botanic Tonics, Inc. (Case filed 2023, ongoing).
[4] Reddit Community “r/QuittingFeelFree.” User reports of dependence and withdrawal (2023–2025).
[5] Centers for Disease Control and Prevention (CDC). Health Effects of Kratom and Kava Use (2024 Update).
Addiction Gap is a certified 501(c)(3) nonprofit